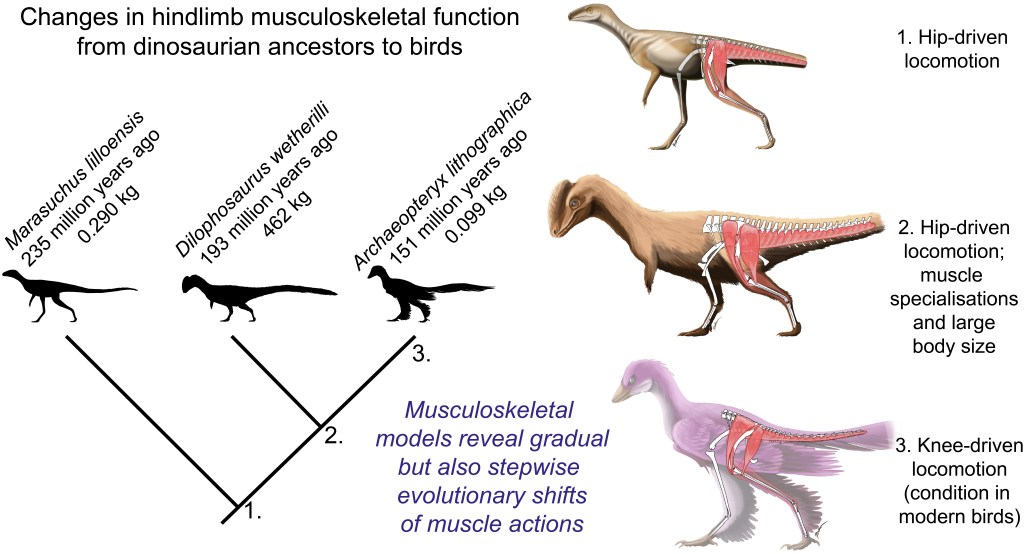
We released a publication that, for me, comes full circle with research that started my career off. Back in 1995 when I started my PhD, I thought it would be great to use biomechanical models and simulations to test how extinct dinosaurs like Tyrannosaurus rex might have moved (or not), taking Jurassic Park CGI animations (for which the goal was to look great) into a more scientific realm (for which the goal is to be “correct”, even at the cost of beauty). “It would be great”, or so I thought, haha. I set off on what has become a ~26 year journey where I tried to build the evidence needed to do so, at each step trying to convince my fairly sceptical mind that it was “good enough” science. For my PhD I mainly focused on reconstructing the hindlimb muscles and their evolution, then using very simple “stick figure”, static biomechanical models of various bipeds to test which could support fast running with their leg muscles, culminating in a 2002 Nature paper that made my early career. I since wrote a long series of papers with collaborators to build on that work, studying muscle moment arms, body/segment centre of mass, and finally a standardized “workflow” for making 3D musculoskeletal models. And gradually we worked with many species, mostly living ones, to simulate walking and running and estimate how muscles controlled observed motions and forces from experiments. This taught us how to build better models and simulations. Now, in 2021, our science has made the leap forward I long hoped for, and the key thing for me is that I believe enough of it is “good enough” for me, which long held me back. This is thus my personal perspective. We have a press release that gives the general story for public consumption; here I’ve written for more of a sciencey audience.

Stomach-Churning Rating: 1/10: just digital muscles.
Earlier in 2021, we simulated tinamou birds in two papers (first one here), the second one revealing our first ever fully predictive simulations, of jumping and landing; detailed here and with a nice summary article here. That research was led by DAWNDINOS postdoc Peter Bishop and featuring new collaborators from Belgium, Dr. Antoine Falisse and Prof. Friedl De Groote. Thanks to the latter duo’s expertise, we used what is called direct collocation (optimal control) simulation; which is faster than standard “single-shooting” forward dynamic simulation. The simulations also were fully three-dimensional, although with some admitted simplifications of joints and the foot morphology; much as even most human simulations do. The great thing about predictive simulations is that, unlike tracking/inverse simulations (all of my prior simulation research), it generates new behaviours, not just explaining how experimentally observed behaviours might have been generated by neuromuscular control.
OK, so what’s this new paper really about and why do I care? We first used our tinamou model to predict how it should walk and sprint, via some basic “rules” of optimal control goals. We got good results, we felt. That is the vital phase of what can be called model “validation”, or better termed “model evaluation”; sussing out what’s good/bad about simulation outputs based on inputs. It was good enough overall to proceed with a fossil theropod dinosaur, we felt.
And so we returned to the smallish Triassic theropod Coelophysis, asking our simulations to find optimal solutions for maximal speed running. We obtained plausible results for both, including compared against Triassic theropod footprints and our prior work using static simulations. Leg muscles acted in ways comparable with how birds use them, for example, and matching some of my prior predictions (from anatomy and simple ideas of mechanics) of how muscle function should have evolved. The hindlimbs were more upright (vertical; and stiff) as we suspect earlier theropods were; unlike the more crouched, compliant hindlimbs of birds.

We observed that the simulations did clever things with the tail, swinging it side-to-side (and up-down) with each step in 3D; and in-phase with each leg: as the leg moved backward, the tail moved toward that leg’s side. With deeper analyses of these simulations, we found that this tail swinging conserved angular momentum and thus mechanical energy; making locomotion effectively cheaper, analogous to how humans swing their arms when moving. This motion emerged just from the physics of motion (i.e., the “multi-body dynamics”); not being intrinsically linked to muscles (e.g. the big caudofemoralis longus) or other soft tissues/neural control constraints (i.e., the biology). That is a cool finding, and because Coelophysis is a fairly representative theropod in many ways (bipedal, cursorial-limb-morphology, big tail, etc.), these motions probably transfer to most other fully bipedal archosaurs with substantial tails. Curiously, these motions seem to be opposite (tail swings left when right leg swings backward) in quadrupeds and facultatively bipedal lizards, although 3D experimental data aren’t abundant for the latter. But then, it seems beavers do what Coelophysis did?

The tail motions, and the lovely movies that our simulations produce, are what the media would likely focus on in telling the tale of this research, but there’s much more to this study. The tinamou simulations raise some interesting questions of why certain details didn’t ideally reflect reality: e.g., the limbs were still a little too vertical, a few muscles didn’t activate at the right times vs. experimental data, the foot motions were awkward, and the forces in running tended to be high. Some of these have obvious causes, but others do not, due to the complexity of the simulations. I’d love to know more about why they happen; wrong outputs from such models can be very interesting themselves.
Speaking of wrong, in order to make our Coelophysis walk and run, we had to take two major shortcuts in modelling the leg muscles. The tinamou model had standard “Hill-type” muscles that almost everyone uses, and they’re not perfect models of muscle mechanics but they are a fair start; it also had muscle properties (capacity for force production, length change, etc.) that were based on empirical (dissection, physiology) data. Yet for our fossil, because we don’t know the lengths of the muscle fibres (active contractile parts) vs. tendons (passive stretchy bits), we adopted a simplified “muscle” model that combined both into one set of properties rather than more realistically differentiating them. It was incredibly important, then, that we try this simple muscle model with our tinamou to see how well it performed; and it did OK but still not “perfect”, and that simple muscle model might not work so well in other behaviours. That was the first major shortcut. Second, again because we don’t know the detailed architecture of the leg muscles in Coelophysis, we had to set very simple capacities for muscular force production: all muscles could only produce at most 2.15 body weights of force. This assumption worked OK when we applied it to our simulation of sprinting in the tinamou (vs. average 1.95 body weights/muscle in the real bird), so it was sufficiently justifiable for our purposes. In current work, we’re examining some alternative approaches to these two shortcuts that hopefully will improve outputs while maximising realism and objectivity.
If you pay close attention, our simulations of Coelophysis output rather high leg-forces, and it’s unclear if that’s due to the simple muscle model, the simple foot modelling, or is actually realistic due to the more vertical (hence stiff) hindlimbs; or all of these. Another intriguing technical finding was that shifting the body’s centre of mass forwards slowed down the simulation’s running speed, as one might expect from basic mechanics (greater leg joint torques), but unlike some prior simulations by other teams.
Users of models and simulations are very familiar with catchphrases like “all models are wrong, but some are useful” or the much more cynical (or ignorant) “garbage in, garbage out”; or the very dangerous attitude that “if the mathematics is correct, then the models can’t be that wrong” (but if the biology is wrong, fuggedaboutit!). These are salutary cautionary tales and catechisms that keep us on our toes, because the visual realism that realistic-looking simulations produce can seduce you into thinking that the science is better than it is. It’s not a field that’s well-suited for those fearful of being wrong. I’ll never think these outputs are perfect; that is a crazy notion; but today I feel pretty good. This was a long time coming for me, and it is satisfying to get to this stage where we can push forwards in some new directions such as comparing simulations of different species to address bigger evolutionary questions.
The wrestling with scepticism never ends, but we can make progress while the match goes on.